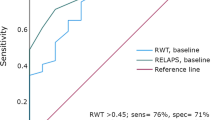
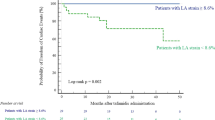
Abstract
Objectives
To analyze clinical predictors of mortality in wild-type transthyretin amyloidosis (wt-ATTR).
Methods
In total, 191 patients (73.8 ± 0.5 years; 176 males, 15 females) with histologically proven wt-ATTR amyloidosis and genetic exclusion of a transthyretin gene variant were included. Comprehensive clinical characteristics, ECG, biomarkers, and echocardiography were analyzed retrospectively. Strain analyses were performed offline using TomTec Imaging Systems, Germany. Univariable and multivariable analyses predicting all-cause mortality were carried out.
Results
Patients presented with significant heart failure (NYHA 2.5 ± 0.8; NT-proBNP 3644 (4981) pg/ml; LV ejection fraction 45.8 ± 15.0%). LogNT-proBNP correlated with indicators of disease severity. Similar results were obtained for basal and midventricular, but not apical longitudinal strain. During median follow-up of 26.2 ± 1.7 months 46 (25.5%) patients died (40 males, 23%; six females, 40%). In female patients 1-/2-year survival was lower [92.9/67.7%; median survival 30.6 (21.1–40.1) months] when compared to male patients [96.5%/86.6%; median survival 63.9 (45.8–82.0) months]. Parameters associated with survival were NT-proBNP, NYHA class, heart rate, midventricular longitudinal strain, mitral annular plane systolic excursion (MAPSE), Karnofsky Index, systolic blood pressure, estimated glomerular filtration rate. Multivariable analysis revealed MAPSE and NT-proBNP as independent predictors of mortality in the whole cohort and midventricular strain in the subgroup of patients in sinus rhythm.
Conclusions
No sex-specific bias was observed between male and female patients with wt-ATTR regarding age at onset and morphological characteristics. Multivariable analysis revealed MAPSE and NT-proBNP as independent predictors of survival in the whole cohort, whereas midventricular longitudinal strain was the only independent predictor in patients in sinus rhythm.



Similar content being viewed by others
References
Plante-Bordeneuve V (2014) Update in the diagnosis and management of transthyretin familial amyloid polyneuropathy. J Neurol 261:1227–1233
Ando Y, Coelho T, Berk JL et al (2013) Guideline of transthyretin-related hereditary amyloidosis for clinicians. Orphanet J Rare Dis 8:1–18
Dubrey S, Ackermann E, Gillmore J (2015) The transthyretin amyloidoses: advances in therapy. Postgrad Med J 91:439–448
aus dem Siepen F, Bauer R, Aurich M et al (2015) Green tea extract as a treatment for patients with wild-type transthyretin amyloidosis: an observational study. Drug Des Devel Ther 9:6319–6317
Dahlem K, Michels G, Kobe C et al (2017) Diagnosis of cardiac transthyretin amyloidosis based on multimodality imaging. Clin Res Cardiol 106:471–473
Kristen AV, Perz JB, Schonland SO et al (2007) Rapid progression of left ventricular wall thickness predicts mortality in cardiac light-chain amyloidosis. J Heart Lung Transplant 26:1313–1319
Mussinelli R, Salinaro F, Alogna A et al (2013) Diagnostic and prognostic value of low QRS voltages in cardiac AL amyloidosis. Ann Noninvasive Electrocardiol 18:271–280
Buss SJ, Emami M, Mereles D et al (2012) Longitudinal left ventricular function for prediction of survival in systemic light-chain amyloidosis: incremental value compared with clinical and biochemical markers. J Am Coll Cardiol 60:1067–1076
Dispenzieri A, Kyle RA, Gertz MA et al (2003) Survival in patients with primary systemic amyloidosis and raised serum cardiac troponins. Lancet 361:1787–1789
Dispenzieri A (2004) Serum cardiac troponins and N-terminal pro-brain natriuretic peptide: a staging system for primary systemic amyloidosis. J Clin Oncol 22:3751–3757
Kristen AV, Giannitsis E, Lehrke S et al (2010) Assessment of disease severity and outcome in patients with systemic light-chain amyloidosis by the high-sensitivity troponin T assay. Blood 116:2455–2461
Palladini G (2003) Serum N-terminal pro-brain natriuretic peptide is a sensitive marker of myocardial dysfunction in AL amyloidosis. Circulation 107:2440–2445
Kristen AV, Rinn J, Hegenbart U et al (2014) Improvement of risk assessment in systemic light-chain amyloidosis using human placental growth factor. Clin Res Cardiol 104:250–257
Mohty D, Pradel S, Magne J et al (2017) Prevalence and prognostic impact of left-sided valve thickening in systemic light-chain amyloidosis. Clin Res Cardiol 106:331–340
Rahman JE, Helou EF, Gelzer-Bell R et al (2004) Noninvasive diagnosis of biopsy-proven cardiac amyloidosis. J Am Coll Cardiol 43:410–415
Gertz MA, Comenzo R, Falk RH et al (2005) Definition of organ involvement and treatment response in immunoglobulin light chain amyloidosis (AL): a consensus opinion from the 10th international symposium on amyloid and amyloidosis. Am J Hematol 79:319–328
de Simone G, Devereux RB, Ganau A et al (1996) Estimation of left ventricular chamber. Am J Cardiol 78:801–807
Lang RM, Badano LP, Mor-Avi V et al (2015) Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr 28:1–39
Devereux RB, Reichek N (1977) Echocardiographic determination of left ventricular mass in man. Anatomic validation of the method. Circulation 55:613–618
Carroll JD, Gaasch WH, McAdam KP (2003) Amyloid cardiomyopathy: characterization by a distinctive voltage/mass relation. Am J Cardiol 49:9–13
Schemper M, Smith TL (1996) A note on quantifying follow-up in studies of failure time. Control Clin Trials 17:343–346
Aurich M, Keller M, Greiner S et al. (2016) Left ventricular mechanics assessed by two-dimensional echocardiography and cardiac magnetic resonance imaging: comparison of high-resolution speckle tracking and feature tracking. Eur Heart J Cardiovasc Imaging 17(12):1370–1378
Gillmore JD, Maurer MS, Falk RH et al. (2016) Nonbiopsy diagnosis of cardiac transthyretin amyloidosis. Circulation 133(24):2404–2412
Dungu JN, Anderson LJ, Whelan CJ, Hawkins PN (2012) Cardiac transthyretin amyloidosis. Heart 98:1546–1554
Meyer S, Teerlink JR, Metra M et al (2017) Sex differences in early dyspnea relief between men and women hospitalized for acute heart failure: insights from the RELAX-AHF study. Clin Res Cardiol 106:280–292
Ogah O, Davison BA, Sliwa K et al (2015) Gender differences in clinical characteristics and outcome of acute heart failure in sub-Saharan Africa: results of the THESUS-HF study. Clin Res Cardiol 104:481–490
Meyer S, Brouwers FP, Voors AA et al (2015) Sex differences in new-onset heart failure. Clin Res Cardiol 104:342–350
Nakagawa M et al (2016) Carpal tunnel syndrome: a common initial symptom of systemic wild-type ATTR (ATTRwt) amyloidosis. Amyloid 23:58–63
Longhi S, Guidalotti PL, Quarta CC et al (2014) Identification TTR-related subclinical amyloidosis with 99mTc-DPD scintigraphy. JACC Cardiovasc Imag 7:531–532
Gonçalves I, Alves CH, Quintela T et al (2008) Transthyretin is up-regulated by sex hormones in mice liver. Mol Cell Biochem 317:137–142
Gorcsan J, Tanaka H (2011) Echocardiographic assessment of myocardial strain. J Am Coll Cardiol 58:1401–1413
Abraham TP, Dimaano VL, Liang HY (2007) Role of tissue Doppler and strain echocardiography in current clinical practice. Circulation 116:2597–2609
Bellavia D, Abraham TP, Pelikka PA et al (2007) Detection of left ventricular systolic dysfunction in cardiac amyloidosis with strain rate echocardiography. J Am Soc Echo 20:1194–1202
Bellavia D, Pellikka PA, Abraham TP et al (2008) Evidence of impaired left ventricular systolic function by Doppler myocardial imaging in patients with systemic amyloidosis and no evidence of cardiac involvement by standard two-dimensional and Doppler echocardiography. Am J Cardiol 101:1039–1045
Koyama J (2003) Longitudinal myocardial function assessed by tissue velocity, strain, and strain rate tissue Doppler echocardiography in patients with AL (Primary) cardiac amyloidosis. Circulation 107:2446–2452
Xylomenos G, Matzaraki V, Papoutsidakis N et al (2015) Myocardial deformation imaging unmasks subtle left ventricular systolic dysfunction in asymptomatic and treatment-naïve HIV patients. Clin Res Cardiol 104:975–981
Geyer H, Caracciolo G, Abe H et al (2010) Assessment of myocardial mechanics using speckle tracking echocardiography: fundamentals and clinical applications. J Am Soc Echocardiogr 23:351–369
Quarta CC, Solomon SD, Uraizee I et al (2014) Left ventricular structure and function in transthyretin-related versus light-chain cardiac amyloidosis. Circulation 129:1840–1849
Phelan D, Collier P, Thavendiranathan P et al (2012) Relative apical sparing of longitudinal strain using two-dimensional speckle-tracking echocardiography is both sensitive and specific for the diagnosis of cardiac amyloidosis. Heart 98:1442–1448
Ternacle J, Bodez D, Guellich A et al (2016) Causes and consequences of longitudinal LV dysfunction assessed by 2D strain echocardiography in cardiac amyloidosis. JACC Cardiovasc Imag 9:126–138
Senapati A, Sperry BW, Grodin JL et al (2016) Prognostic implication of relative regional strain ratio in cardiac amyloidosis. Heart 102:748–754
Shi J, Guan J, Jiang B et al (2010) Amyloidogenic light chains induce cardiomyocyte contractile dysfunction and apoptosis via a non-canonical p38 MAPK pathway. Proc Nat Acad Sci 107:4188–4193
Takemura G, Takatsu Y, Doyama K et al (1998) Expression of atrial and brain natriuretic peptides and their genes in hearts of patients with cardiac amyloidosis. J Am Coll Cardiol 31:254–265
Acknowledgements
This study was supported by the German Center for Cardiovascular Research (DZHK) without any financial funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Financial disclosures.
Nothing to declare. No Grants, contracts or other forms of financial support.
Rights and permissions
About this article
Cite this article
Siepen, F.a.d., Bauer, R., Voss, A. et al. Predictors of survival stratification in patients with wild-type cardiac amyloidosis. Clin Res Cardiol 107, 158–169 (2018). https://doi.org/10.1007/s00392-017-1167-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00392-017-1167-1